|
9/6/2023 0 Comments Molar mass of carbon dioxide 
The following thermodynamic properties will be calculated:ĭensity, dynamic viscosity, kinematic viscosity, specific enthalpy, specific entropy, specific isobar heat capacity cp, specific isochor heat capacity cp, thermic conductivity, coefficient of thermal expansion, heat conductance, thermal diffusivity, Prandtl-number, coefficient of compressibility Z, speed of sound.Ĭalculation of carbon dioxide : if you found an error, please mail to: No garanty for correctness. Lower limit for calculation: -55 C, 5,4 bar bar upper limit: 30 C, 72,14 bar. Lower limit for calculation: -55 C, 1 bar upper limit: 900 C, 1000 barĬalculation of thermodynamic state variables of carbon dioxide at saturation state, boiling curve If given the molar mass (how many g/mol of the compound) then you can calculate the. The mass of a nitrogen molecule is 4.68 1026 kg. Equation 20.18 can be used to find the rms velocity given the temperature, Boltzmanns constant, and the mass of the atom or molecule. Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have.Email: scientific and engineering data onlineĬalculation of thermodynamic state variables of carbon dioxide For carbon dioxide, the molecular formula is CO2, and the empirical. The mass of a nitrogen molecule is 4.68 1026 kg. 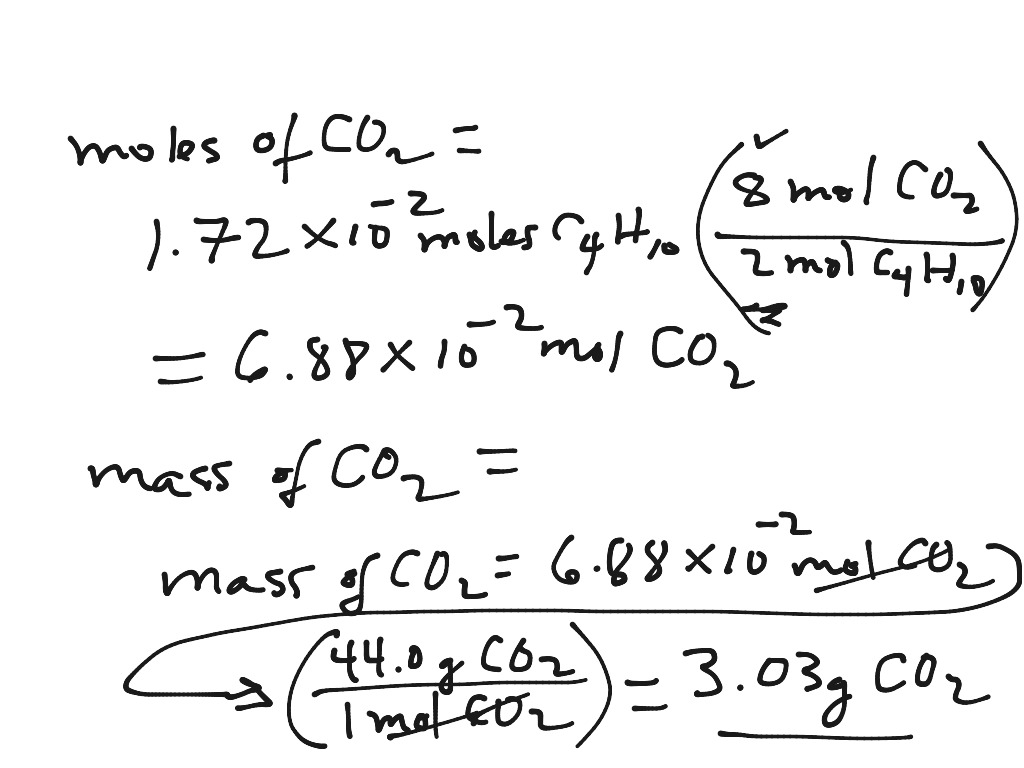
In humans, this carbon dioxide is carried through the venous system and is breathed out through the lungs, resulting in lower concentrations in the arteries. Carbon dioxide is an acidic gas which releases during respiration. A list of some molar masses can be seen below. Content The body produces approximately 2.3 pounds (1.0 kg) of carbon dioxide per day per person, containing 0.63 pounds (290 g) of carbon. The main GHGs in the Earths atmosphere are water vapour, carbon dioxide (CO2), methane (CH4), nitrous oxide (N2O) and ozone. Molecular mass is the sum of the masses of all the individual atoms present in a molecule or compound. The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. The molar mass of glucose is 180.15 g/mol,180.15 g/mol, the molar mass of ethanol is 46.08 g/mol,46.08 g/mol, and the molar mass of carbon dioxide is 44.01 g/mol.44.01 g/mol. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. Calculate the average molar mass from your three trials. Molar mass of CO: Calculate the experimental molar mass of carbon dioxide for each trial using the ideal gas law. We assume you are converting between grams CO2 and mole.You can view more details on each measurement unit: molecular weight of CO2 or mol This compound is also known as Carbon Dioxide.The SI base unit for amount of substance is the mole. Mass of CO2: Calculate the mass of carbon dioxide in the flask for each trial from part 3 of the procedure and part 3 of the calculations. How many grams CO2 in 1 mol The answer is 44.0095. First we find the molar mass for Carbon dioxide using the Periodic Table. More information from the unit converter. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects There are two steps to find the mass of a single molecule of Carbon dioxide (CO2). However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). 
Here, we got 88 'g' of carbon dioxide, and we need to convert it into moles. For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. We have the balanced equation (without state symbols): 6H2O+6CO2->C6H12O6+6O2 So, we would need six moles of carbon dioxide to fully produce one mole of glucose. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
